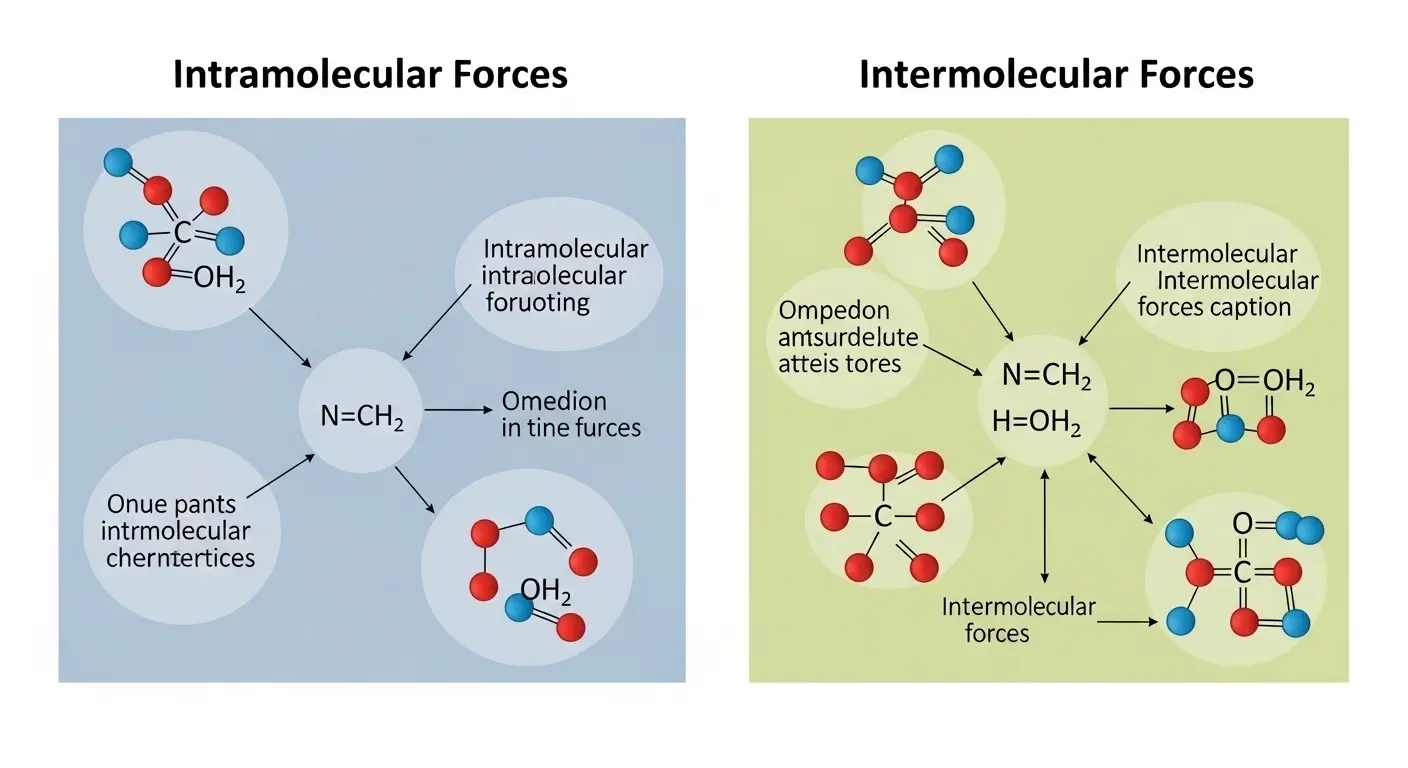
Imagine holding a brick in your hand. The strength that keeps the brick solid and intact comes from forces within it, while the slight attraction between different bricks stacked together helps form a wall. This simple example mirrors the difference between intramolecular and intermolecular forces in chemistry.
Intramolecular forces act within a molecule, holding atoms together, while intermolecular forces act between molecules, influencing how they interact with one another.
Understanding the difference between intramolecular and intermolecular forces is essential because these forces define how substances behave in everyday life from water boiling to ice melting.
The difference between intramolecular and intermolecular interactions explains why some materials are strong while others are easily separated. In fact, the difference between intramolecular and intermolecular forces plays a key role in chemistry, biology, and even industrial applications.
Pronunciation
- Intramolecular
- US: /ˌɪn.trə.məˈlek.jə.lɚ/
- UK: /ˌɪn.trə.məˈlek.jʊ.lə/
- Intermolecular
- US: /ˌɪn.t̬ɚ.məˈlek.jə.lɚ/
- UK: /ˌɪn.tə.məˈlek.jʊ.lə/
Difference Between Intramolecular and Intermolecular
1. Definition
- Intramolecular: Forces within a molecule.
Examples:- Covalent bond in water (H₂O)
- Ionic bond in sodium chloride (NaCl)
- Intermolecular: Forces between molecules.
Examples:- Hydrogen bonding between water molecules
- Van der Waals forces in gases
2. Strength
- Intramolecular: Strong forces.
Examples:- Carbon-carbon bonds in diamond
- Peptide bonds in proteins
- Intermolecular: Weak forces.
Examples:- Dipole-dipole interactions
- London dispersion forces
3. Function
- Intramolecular: Holds atoms together in molecules.
Examples:- Formation of glucose
- DNA backbone structure
- Intermolecular: Holds molecules together.
Examples:- Water cohesion
- Liquid formation
4. Energy Requirement
- Intramolecular: Requires high energy to break.
Examples:- Breaking O-H bond
- Splitting CO₂ molecule
- Intermolecular: Requires low energy.
Examples:- Boiling water
- Evaporation of alcohol
5. Type of Bonds
- Intramolecular: Covalent, ionic, metallic.
Examples:- Metallic bond in iron
- Covalent bond in methane
- Intermolecular: Hydrogen bonds, dipole forces.
Examples:- Hydrogen bonding in ammonia
- Dipole interaction in HCl
6. State Influence
- Intramolecular: Determines chemical identity.
Examples:- Water vs hydrogen peroxide
- Methane vs ethane
- Intermolecular: Determines physical state.
Examples:- Ice vs liquid water
- Gas vs liquid nitrogen
7. Occurrence
- Intramolecular: Inside molecules only.
Examples:- Bond in oxygen molecule
- Bond in glucose
- Intermolecular: Between molecules.
Examples:- Attraction between water molecules
- Attraction in liquid CO₂
8. Role in Chemical Reactions
- Intramolecular: Broken/formed in reactions.
Examples:- Combustion reaction
- Photosynthesis
- Intermolecular: Usually unchanged.
Examples:- Dissolving sugar
- Mixing liquids
9. Stability
- Intramolecular: Provides molecular stability.
Examples:- Stable benzene ring
- Stable DNA strands
- Intermolecular: Provides temporary attraction.
Examples:- Surface tension
- Temporary clustering
10. Examples in Daily Life
- Intramolecular: Cooking chemical reactions.
Examples:- Baking bread
- Burning fuel
- Intermolecular: Physical changes.
Examples:- Melting ice
- Condensation of steam
Nature and Behaviour
- Intramolecular Forces: Strong, stable, and responsible for forming substances. They rarely change unless a chemical reaction occurs.
- Intermolecular Forces: Weak, flexible, and responsible for physical properties like boiling point, melting point, and viscosity.
Why People Are Confused
People often confuse them because both involve “forces” between particles. The key confusion lies in not distinguishing whether the force is inside a molecule (intra) or between molecules (inter).
Table: Difference and Similarity
| Feature | Intramolecular | Intermolecular | Similarity |
| Location | Within molecule | Between molecules | Both involve attraction |
| Strength | Strong | Weak | Both affect matter |
| Function | Forms molecules | Holds molecules together | Important in chemistry |
| Energy | High | Low | Energy dependent |
| Type | Covalent, ionic | Hydrogen, dipole | Both are forces |
Which is Better in What Situation?
Intramolecular forces are “better” when stability and structure are needed. For example, in building strong materials like plastics or metals, strong internal bonding is essential. Without intramolecular forces, molecules would not even exist.
Intermolecular forces are more useful when flexibility and change are required. They allow substances to melt, evaporate, and dissolve. In biological systems, these weak forces enable processes like protein folding and DNA interactions, making life possible.
Metaphors and Similes
- Intramolecular: “Like the glue holding a puzzle together.”
- Intermolecular: “Like a gentle handshake between people.”
Connotative Meaning
- Intramolecular: Positive (strength, stability)
- Example: “A strong bond within a family.”
- Intermolecular: Neutral (interaction, connection)
- Example: “Social connections between communities.”
Idioms or Proverbs
(No direct idioms exist, but adapted usage)
- “Strong bonds build strong foundations.” (Intramolecular idea)
- “Connections make the world go round.” (Intermolecular idea)
Works in Literature
(No direct works named after these terms, but used in science literature)
- Chemical Bonding and Molecular Structure Academic Textbook, 2010
- The Nature of Chemical Bonds Linus Pauling, 1939
Movies Related to Concepts
(Not directly named, but conceptually relevant)
- Interstellar (2014, USA) explores interactions and forces
- The Theory of Everything (2014, UK) scientific concepts
FAQs:
1. What is the main difference?
Intramolecular forces act within molecules, while intermolecular forces act between molecules.
2. Which is stronger?
Intramolecular forces are much stronger.
3. Do intermolecular forces affect boiling point?
Yes, stronger intermolecular forces lead to higher boiling points.
4. Are both important?
Yes, both are essential for chemical and physical properties.
5. Can one exist without the other?
No, molecules need intramolecular forces first before intermolecular forces can act.
How Both Are Useful for Surroundings
Intramolecular forces create the substances we use daily, such as water, plastics, and metals. Intermolecular forces allow these substances to change states, dissolve, and interact, making everyday processes like cooking and breathing possible.
Final Words
Intramolecular forces build the foundation of matter, while intermolecular forces shape how that matter behaves.
Conclusion:
The difference between intramolecular and intermolecular forces lies at the heart of chemistry. Intramolecular forces are strong and responsible for forming molecules, while intermolecular forces are weaker and control physical properties. Both are equally important in understanding how substances exist and interact in the world. From the structure of DNA to the boiling of water, these forces play a critical role in science and everyday life.
By understanding their differences, learners and experts can better grasp chemical behavior and apply it in real-world situations.

I am an English content writer with more than 8 years of experience in writing about English word differences, grammar clarity, and everyday language usage. I am passionate about helping learners avoid common mistakes caused by similar-looking or confusing English words.
At diffrulee.com, I write easy-to-understand guides that explain the difference between commonly confused words, spelling variations, and correct usage with real-life examples. My content is especially useful for students, writers, and non-native English speakers who want quick, accurate, and practical answers.